ERN-EuroBloodNet repository of Clinical Practice Guidelines and Other Clinical Decision Making Tools are searchable at the new website section!
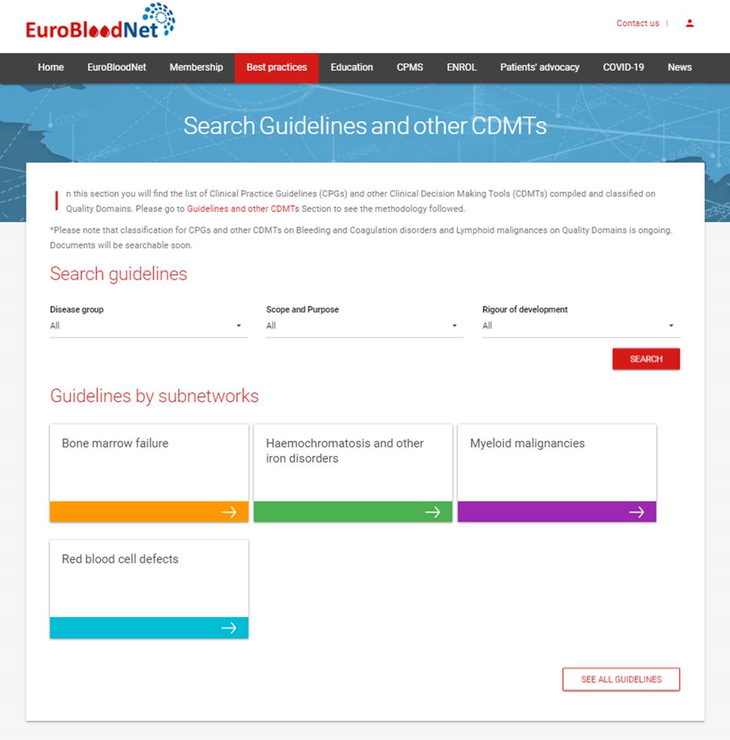
A total of 117 GPGs and other CDMTs have been compiled for the six subnetworks, from which 68 have been classified on Quality Domains. Check now the new repository released, including a search tool for their findability on Subnetwork, Disease group, Scope and Purpose and Rigour of Development approach!
During last years, a number of Clinical Practice Guidelines (CPGs) and other Clinical Decision Making Tools (CDMTs) on RHD have been published at both national or European level. However, up to date, there is no repository of documents including quality assessment, evaluation of gaps or level of implementation, updates or any system to guarantee their usefulness and impact in patients' quality of life.
Accordingly, one of the key objectives established by ERN-EuroBloodNet is to foster best practice sharing in RHD by creating a comprehensive public repository of reliable evidence based CPGs and other CDMTs on RHD, ranging from prevention, diagnostic tests and treatments to the organisation of patient-centred management in multidisciplinary teams. In this context, an strategy was developed for:
- Compilation of existing CPGs and other CDMTs
- Classification on Quality Domains based on:
- Scope and purpose
- Patients' involvement
- Rigour of development:
A: evidence and consensus-based guidelines / recommendations: grading system for strength of recommendation involving assessment of the quality of evidence (study design, study quality, consistency) (e.g. GRADE, SIGN)
B: consensus-based guidelines / recommendations: formal consensus development techniques (e.g. Delphi method, nominal group technique)
C: expert opinion: entirely based on opinions by experts without formal consensus development techniques
As a result, a total of 117 GPGs and other CDMTs have been compiled for the six subnetworks, from which 68 have been classified on Quality Domains, belonging to red blood cell, bone marrow failures, hemochromatosis and iron disorders, and myeloid malignancies subnetworks. Classification for lymphoid malignancies and bleeding and coagulation disorders subnetworks are still ongoing.
We are delighted to announce the launch of the repository of 68 CPGs and other CDMT available at the new dedicated section, including a search tool for their findability on Subnetwork, Disease group, Scope and Purpose and Rigour of Development approach! Check it now and discover the ERN-EuroBloodNet central platform for sharing best practices!
