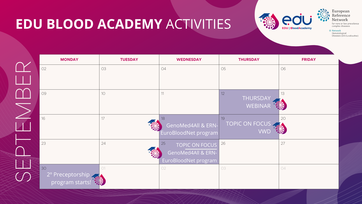
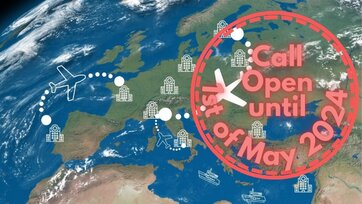
ERN-EuroBloodNet is officially supporting partner of the World Orphan Drug Congress Europe!
2025-08-26
Save the date! This is the largest and most established event globally for orphan drugs and rare diseases.
Read More